Every provider who performs CoolSculpting should talk about the potential for negative side effects even if rare. This includes paradoxical adipose hyperplasia, or PAH. Most providers don’t mention it ahead of time. The incidence is low, fewer than 0.033% of treatments, or 1 in 3000, and many clinics consider it too rare to mention. But rare isn’t the same as nonexistent, and informed consent isn’t a selective disclosure. Get clarity on CoolSculpting side effects here.
This article explains what PAH is, how often it occurs, why it happens, and what the treatment pathway looks like if it does. If you’re evaluating CoolSculpting as an option, the providers who are transparent about the full risk profile, including the edges, are the ones worth trusting with your body. For the complete safety picture, including FDA clearance and common side effects, see Is CoolSculpting Safe? Medical Risks & FDA Clearance.
What Paradoxical Adipose Hyperplasia Actually Is
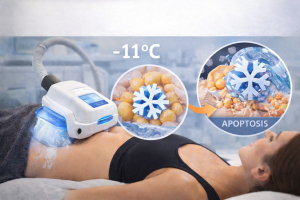
Paradoxical adipose hyperplasia, PAH, is a complication in which the treated fat area enlarges instead of shrinking. The word “paradoxical” is precise: the expected response to cryolipolysis is fat cell death and volume reduction. In PAH, the opposite occurs. The fat cells in the treated zone will increase in volume, creating a firm, visible mass roughly in the shape of the CoolSculpting applicator.
PAH typically becomes noticeable 2 to 3 months after treatment, the same window when normal results would be developing. Instead of gradual contour improvement, the area develops a distinct area of fullness. The mass is firm to the touch, clearly defined, and does not resolve on its own. Research indexed in PubMed has documented the clinical presentation and confirmed that the enlarged tissue requires intervention rather than watchful waiting.
The mechanism behind PAH is not fully understood. The leading hypothesis is that in a small subset of patients, the cryolipolysis stimulus triggers fat cell proliferation or hypertrophy rather than apoptosis. Why certain patients respond this way while the vast majority undergo normal apoptotic clearance remains an active area of investigation. What is well established is the incidence rate, the presentation, and the corrective pathway.
How Rare Is PAH, and Who Is at Risk
Published adverse event data places PAH incidence at fewer than 0.033% of CoolSculpting treatments. For context, that’s roughly 1 in every 3,000 treatment cycles. Across over 17 million CoolSculpting treatments performed globally, PAH remains a statistically rare event. PAH entered the public conversation after supermodel Linda Evangelista shared her experience, an example of why rare risks still deserve upfront discussion.
The American Society for Dermatologic Surgery includes PAH in its cryolipolysis complication literature, classifying it as a known but infrequent adverse outcome. No reliable predictor has been identified. PAH does not correlate with treatment area, applicator type, number of cycles, or patient demographics in any consistent pattern documented to date. It can occur in any patient, on any cycle, in any area.
This unpredictability is exactly why disclosure matters. A provider cannot screen for PAH the way they screen for candidacy. There is no tissue test, no biomarker, no risk profile that reliably identifies who will develop it. The only responsible approach is to ensure every patient knows it exists before treatment begins and that roughly 1 in 3000 have seen it. Knowing the potential for CoolSculpting side effects, such as PAH, is necessary.
Why Most Providers Don’t Talk About PAH
The clinical rationale for not discussing PAH is straightforward: the incidence is so low that many providers consider it negligible. Some have performed thousands of cycles without encountering a single case. From a purely statistical standpoint, PAH is one of the least likely complications a CoolSculpting patient will ever face.
But informed consent is not a statistical exercise. The Mayo Clinic frames informed consent as a process in which patients receive information about all known risks, benefits, and alternatives, so they can make decisions aligned with their own values and risk tolerance. Omitting a known complication because it’s improbable doesn’t meet that standard.
The providers who skip PAH disclosure are often the same ones who simplify the rest of the process: abbreviated consultations, minimal candidacy screening, single-cycle treatment plans without follow-up. Transparency on rare risks is a signal of how a practice approaches the entire patient relationship. If a clinic won’t tell you about a 1-in-3000 risk, the question becomes what else they’re simplifying. To see how provider differences affect outcomes beyond safety, read Does CoolSculpting Really Work? Clinical Outcomes.
What Happens If PAH Occurs: CoolSculpting Rare Side Effects
PAH does not resolve spontaneously. The enlarged tissue is persistent, and no non-surgical approach has demonstrated reliable reversal. The standard corrective treatment is liposuction to remove the hyperplastic fat from the affected area.
The corrective timeline varies, but patients are typically advised to wait 6 to 9 months after the onset of PAH before pursuing surgical correction. This allows the tissue to stabilize so the surgeon can achieve a complete and even removal. The liposuction procedure itself is targeted and confined to the PAH-affected zone; it is not a large-scale surgical event.
PAH is cosmetic, not dangerous. It does not indicate a systemic problem, does not affect organ function, and does not create long-term health risks beyond the visual change. The discomfort is almost entirely psychological and aesthetic: patients expected improvement and received the opposite. That emotional weight is real, and it is compounded when the patient was never told the risk existed.
How Sculptology’s Process Addresses PAH Risk
PAH cannot be predicted or prevented with current clinical knowledge. What can be controlled is disclosure, monitoring, and response.
At Sculptology, PAH is part of every informed consent conversation. Every patient who consults for CoolSculpting Elite is told the incidence rate, shown what PAH looks like, given the timeline for identification, and walked through the corrective pathway before any treatment decision is made. Our clinical team reviews candidacy assessments and ensures the consent process meets clinical standards, not just legal minimums.
Sculptology’s structured follow-up schedule, check-ins at 4, 8, and 12 weeks with S-Index tracking, serves a dual purpose. It tracks expected contour progress, and it provides clinical checkpoints where PAH would be identified early if it occurred. The Comfort Call on day 4 post-treatment is designed to manage late-onset nerve sensitivity, but it also opens a communication line during the window when patients are most likely to have questions or concerns about their body’s response.
No provider can guarantee PAH won’t occur. The S-Method™ doesn’t eliminate the risk; no process can. What it does is ensure that patients are informed before treatment, monitored after treatment, and supported through the corrective pathway if PAH is ever identified. That’s the difference between a practice that manages risk and one that pretends it doesn’t exist.
| UNIQUE SCULPTOLOGY INSIGHT Why Transparency Is the Actual Safety Net The question isn’t whether CoolSculpting side effects exist; every procedure has them. It’s whether your provider structured a process that accounts for them before, during, and after treatment. This means the real differentiator in CoolSculpting safety isn’t the incidence rate of any single complication. It’s whether the practice built informed consent, structured monitoring, and a clinical escalation pathway into the standard of care, or whether they treated disclosure as optional. Apoptosis at -11°C is selective: fat cells crystallize while surrounding tissue remains unaffected. That thermal precision is what makes the procedure safe for the vast majority of patients. But precision in the mechanism doesn’t replace precision in the process around it. |
Frequently Asked Questions
What is paradoxical adipose hyperplasia (PAH)?
Paradoxical adipose hyperplasia is a rare complication of cryolipolysis in which the treated fat area enlarges rather than shrinks. Instead of undergoing apoptosis, the affected fat cells increase in volume, creating a firm, visible mass in the shape of the applicator. PAH occurs in fewer than 0.033% of CoolSculpting treatments.
How common is PAH after CoolSculpting?
Published adverse event data places the incidence of PAH at fewer than 0.033% of treatments, roughly 1 in every 3,000 cycles. While rare, it is a real complication that is discussed at Sculptology during an important informed consent process before treatment.
Can PAH be treated or reversed?
Yes. PAH is treatable. The standard corrective approach is liposuction to remove the enlarged tissue. PAH does not resolve on its own and typically requires surgical intervention, but the condition is cosmetic and does not pose a health risk, yet has a visual change.
Does Sculptology disclose PAH risk before treatment?
Yes. Sculptology includes PAH disclosure in every informed consent process. Every patient is told the incidence rate, what PAH looks like, the timeline for identification, and the corrective pathway before any treatment begins. Transparency on rare risks is part of the clinical standard.
What are the most common CoolSculpting side effects?
The most common CoolSculpting side effects are temporary and localized: numbness, redness, swelling, bruising, and mild discomfort in the treated area. Late-onset nerve sensitivity can occur 3 to 5 days post-treatment. These effects typically resolve within days to weeks without intervention.
Why do some providers not mention PAH?
Some providers avoid discussing PAH because the incidence is so low that they consider it unlikely to affect their patients, or because they fear the disclosure will discourage treatment. Clinical ethics and informed consent standards require that all known risks, regardless of probability, be communicated to patients before any elective procedure, a practice that Sculptology provides.
Related Reading
Is CoolSculpting Safe? Medical Risks & FDA Clearance
Does CoolSculpting Really Work? Clinical Outcomes
Ready to See What The S-Method Can Do?
Sculptology offers complimentary consultations for CoolSculpting Elite and The S-Method™ transformation process.
Book your consultation online:
sculptology.com/book-consultation
Or call our Lafayette location:
(925)329-6333
Located in Lafayette, California, serving the East Bay and greater San Francisco area.