If you’re researching CoolSculpting safety, you’re asking the right question at the right time. Before committing to any body contouring treatment, understanding the clinical evidence, not the sales offers, is what separates an informed decision from a hopeful one.
CoolSculpting Elite has been FDA-cleared for 9 body areas since 2010, and over 17 million treatments have been performed globally. Those numbers reflect a technology with a well-documented safety record. But CoolSculpting safety isn’t just about the device; it’s also about the process surrounding it, the provider operating it, and whether you’re the right candidate in the first place.
What FDA Clearance Actually Means for CoolSculpting Safety
There’s a common misunderstanding worth correcting: CoolSculpting is FDA-cleared, not FDA-approved. These are different regulatory pathways. Clearance means the device underwent clinical trials demonstrating safety and efficacy, and the FDA reviewed that evidence before authorizing it for specific uses.
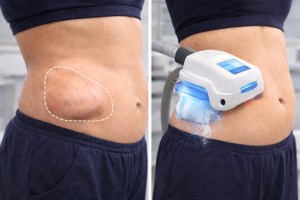
CoolSculpting Elite is cleared for the treatment of visible fat bulges in nine body areas. These cleared areas include the abdomen, flanks, inner thighs, outer thighs, distal thigh, chin (including the submental and submandibular area), upper arms, back, and the axillary area surrounding the upper back. That clearance is based on the mechanism of cryolipolysis, controlled cooling that targets fat cells while leaving surrounding tissue unaffected. The American Society for Dermatologic Surgery recognizes cryolipolysis as an established, evidence-based approach to non-surgical fat reduction, reflecting professional consensus that extends beyond any single manufacturer’s claims.
What clearance doesn’t mean: it doesn’t guarantee a specific outcome for every patient. It confirms that the technology is safe and effective when used as intended, by qualified providers, on appropriate candidates. Those three conditions matter more than most people realize.
Common Side Effects and What to Expect
The most frequently reported CoolSculpting side effects are temporary and localized to the treated area. They include numbness, redness, swelling, bruising, tingling, and mild discomfort. For most patients, these resolve within days to weeks. They are expected physiological responses to controlled cooling, not complications.
Some patients experience late-onset nerve sensitivity, which typically appears 3-5 days after treatment. This is a known response and, at Sculptology, we proactively manage it through a structured follow-up process. What the science shows, and what we’ve confirmed in practice, is this: temporary side effects are common, predictable, and self-limiting.
CoolSculpting is considered one of the lowest-risk treatments available in the non-invasive fat reduction category, and everything in its side effect profile is self-resolving, no ongoing management and no secondary procedures for the standard effects.
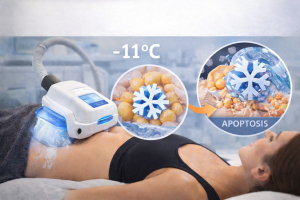
The clinical distinction that matters: cryolipolysis triggers apoptosis (programmed cell death), not necrosis (traumatic cell rupture). Apoptotic cells are metabolized through the lymphatic system gradually over weeks. This mechanism is why the side effect profile is mild; the body is clearing dead cells naturally through normal metabolic pathways, not managing tissue trauma.
The Rare Risk: PAH
Transparency requires addressing the risks that many providers avoid acknowledging. Paradoxical adipose hyperplasia (PAH) is a condition where the treated fat area enlarges instead of shrinking, a factor in CoolSculpting safety. Published adverse event data show a PAH incidence rate of less than 0.033%. That means fewer than about 1 in 3,000 patients have experienced PAH.
PAH is rare, but it’s real. It typically appears 2-6 months after treatment and presents as a firm, defined enlargement in the treated zone. It is treatable, usually through liposuction, but it requires acknowledgment of the risk upfront, not after the fact.
At Sculptology, we acknowledge the importance of CoolSculpting safety and the low PAH risk with every patient during the consultation. We believe informed consent means addressing the full clinical picture, including the outcomes that are statistically unlikely but personally significant if they occur. For a deeper look at PAH, what causes it, how it presents, and how it’s managed, read our dedicated article: Understanding PAH: Transparency on Side Effects.
Why Candidacy Is a Safety Decision
Not everyone is a good candidate for CoolSculpting, and candidacy screening is where safety starts, before the device is ever turned on. CoolSculpting treats subcutaneous fat (the pinchable layer under the skin), not visceral fat (the deeper fat surrounding internal organs). Treating the wrong type of fat doesn’t just produce poor results; it creates a dissatisfied patient who blames the technology for what is essentially a screening failure.
The clinical criteria is specific. Good candidates who are at or near their goal weight with localized areas of pinchable fat that haven’t responded to diet and exercise. At Sculptology, every consultation begins with the Bio-Mechanical Survey, a clinical assessment that includes a tissue laxity test, the Pinch Metric (minimum one inch of pinchable subcutaneous fat), and a diastasis recti screen. This process exists to protect you from a treatment that isn’t right for your body, and peer-reviewed research published on PubMed consistently supports candidacy screening as a primary factor in both safety outcomes and patient satisfaction.
People with cryoglobulinemia, cold agglutinin disease, or paroxysmal cold hemoglobinuria should not receive CoolSculpting. In plain terms, these are rare cold-sensitive blood disorders where exposure to cold can trigger abnormal blood reactions, so CoolSculpting isn’t appropriate. These conditions are screened during the medical history review, another reason physician oversight matters.
What Physician Oversight Changes About CoolSculpting Safety
CoolSculpting safety isn’t only a result of the device. It’s also a function of who evaluates you, who maps your treatment, and who manages the process from start to finish.
Sculptology’s team of providers includes members of a CoolSculpting national training team with over 10+ years of experience working with the technology. They don’t just perform CoolSculpting; they train other providers on protocols and safety standards. With over eight years of experience with the technology and a completion rate of over 25,000 treatments, this specialization creates a clinical advantage that generalist providers do not have. You see the same provider at Sculptology throughout your entire treatment plan. Consistency in who evaluates, maps, and executes your treatment is optimal at Sculptology.
This matters because the variables that affect CoolSculpting safety, candidate selection, treatment mapping, post-treatment massage technique, and metabolic spacing between sessions are all managed by the provider, not the machine. A CoolSculpting Elite treatment performed under physician oversight with clinical candidacy screening is a fundamentally different safety level than the same device operated without that infrastructure. The device is standardized. The customized process around it is not.
| UNIQUE SCULPTOLOGY INSIGHT Why the Safety Question Is Really a Provider Question The question isn’t whether CoolSculpting is safe. It’s whether the process surrounding it is built to improve safety. The real variable isn’t the FDA clearance, the device model, or the treatment temperature. It’s the candidacy screen that happens before any of that, the clinical assessment that determines whether your anatomy, your fat type, and your health history make you an appropriate candidate. At Sculptology, the Bio-Mechanical Survey exists for exactly this reason. Thermal gradient specificity at -11°C ensures fat cells are targeted while surrounding tissue is protected, but only if the right tissue is being treated in the first place by our experienced providers. |
Frequently Asked Questions
Is CoolSculpting FDA-cleared or FDA-approved?
CoolSculpting Elite is FDA-cleared, which is the correct regulatory term for medical devices. FDA clearance means the device has been reviewed for safety and efficacy through clinical trials. It has been FDA-cleared for nine body areas since 2010.
What are the most common CoolSculpting side effects?
The most common side effects are temporary and localized: numbness, redness, swelling, bruising, and mild discomfort in the treated area. These typically resolve within days to weeks. They are expected responses to controlled cooling, not signs of a larger problem.
What is paradoxical adipose hyperplasia, and how common is it?
Paradoxical adipose hyperplasia (PAH) is a rare adverse event where the treated fat area enlarges instead of shrinking. Published adverse event data show a PAH incidence rate of less than 0.033%, one in 3000 people. It is treatable, and an experienced provider will discuss this risk during your consultation.
Does CoolSculpting safety depend on the provider?
Yes. CoolSculpting safety is affected by candidacy screening, treatment mapping, and the post-treatment process. A physician-supervised practice that screens with a clinical assessment like the Bio-Mechanical Survey at Sculptology identifies contraindications and poor candidates before treatment begins, which directly reduces risk.
Who should not get CoolSculpting?
CoolSculpting is not appropriate for individuals with cold-sensitive blood disorders (cryoglobulinemia, cold agglutinin disease, or paroxysmal cold hemoglobinuria). It is also not designed for weight loss, although fat reduction occurs, nor for treating visceral fat. A clinical candidacy assessment at Sculptology determines whether CoolSculpting is safe and appropriate for your specific situation.
How does CoolSculpting avoid damaging skin, nerves, and muscle?
Cryolipolysis works because fat cells crystallize at a higher temperature than surrounding tissue. At approximately -11°C, fat undergoes crystallization while skin, muscle, and nerves remain unaffected. This thermal selectivity is the foundation of CoolSculpting’s safety profile and effectiveness.
Related Reading
Does CoolSculpting Really Work? Clinical Outcomes
Understanding PAH: Transparency on Side Effects
Ready to See What The S-Method Can Do?
Sculptology offers complimentary consultations for CoolSculpting Elite and The S-Method™ transformation process.
Book your consultation online:
sculptology.com/book-consultation
Or call our Lafayette location:
(925)329-6333
Located in Lafayette, California – serving the East Bay and greater San Francisco area.